Chlor-Alkali Plant
Process
Friday, July 19, 2019
The reason for Dow Chemical being in Fort Saskatchewan in the first place was dictated firstly by the huge salt beds 6000 feet under ground. Second was access to lots of water, in this case the North Saskatchewan River. And thirdly, local markets for the chlorine and caustic soda produced. Since Alberta and British Columbia have large pulp and paper mills that use both chemicals, Fort Saskatchewan was a no-brainer for a plant site.Send Comments
Friday, July 19, 2019
Lorem ipsum dolor sit amet, consectetur adipisicing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua.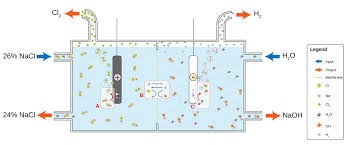
At both ends of the skid were the copper cathodes,
connected by a large collection of about 50 cables,
2" in diameter. These power cables transmitted the
power from the rectifier room where AC was
converted to DC. No small battery here: I remember
80,000 Amperes at 550 volts showing on the control panel. This was for the cells
only, not the entire plant or plant site! At the time I was there, in the 1970’s, Dow
Chemical was the 3rd largest power consumer in the province, only Edmonton and
Calgary used more. When more chlorine production was required, it was a simple
matter of turning the big black dial on the control panel that changed the current flow.
More current caused more electrolysis which created more product.

The cells are not some puny little
things the size of a desk. No, these
behemoths are 12 feet by 12 feet
square, by 2 feet wide, and that’s just
ONE cell! Forty of these together
make up a skid. The tops and bottoms are 12 inch thick concrete and the sides
something like 8 inches thick. Inside each cell is a metal screen, coated with paste
asbestos, and inside the screen was a large 1" thick graphite block (fluffy asbestos
that looks like the loose, gray coloured, cellulose insulation in house attics, is like
papier-mâché when it gets wet. It makes a nice thick paste.) These are the anodes of
what can best be described as a big battery in reverse. Forty of these cells are pushed
together, sealed with big gobs of sticky black "mastic", a cross between rubber and
home caulking. To keep this skid together, 4 of the longest bolts you could ever
imagine squeeze the 4 corners together. It’s like handling a very large Dagwood
sandwich, with your fingers representing the 4 clamps. It’s all put together
"Mechano-style". Think of one of those Betty Crocker square freezer cakes cut up into
1" slices and you basically have the layout and proportions of a skid. The small plant,
where I worked, had 5 of these skids in operation, with one always down for
maintenance or rebuild..
Friday, July 19, 2019
Two ¼" plastic hoses coming off a large 6" header pipe fed brine into each side of the cell. At the bottom of each cell, was a 2" plastic outlet pipe from where the caustic solution flowed, into a large 2 foot wide concrete collecting trough. Each trough fed into an underground storage tank from where the liquid was pumped to the caustic plant (the "alkali" in Chlor-Alkali). Chlorine and hydrogen both came off the top of the center of the skid and could be kept separate by virtue of their different molecular weights, chlorine being much heavier than hydrogen.Chlorine of course is a dangerous gas. In strong concentrations it attacks the tissues of the lungs making breathing difficult, or impossible. In the latter case, you die of asphyxiation. When I would walk in the cell area, there was always a smell of chlorine and a slight haze was obvious. When the haze became green, it was prudent to use the respirator (hanging from my neck at all times). When it was thick enough to really burn your eyes I had to don a Scott Air Pac.
Gate? What Gate?
While working at Chlor-Alkali I got a sore throat (go figure!) so I went to my local doctor in St.Albert. I didn’t talk to the Company nurse or doctor because I didn’t see anyone else from the Chlorine Plant complaining, so assumed this was a "local" thing. The doctor asked where I worked and with what chemicals. As soon as I mentioned the word "chlorine" he said "Well that’s why you have a sore throat!". (Doctors go to school for 10 years to make this quick determination.) He then made me fill in a form for the Workmen’s Compensation Board. A week later, the Safety Supervisor who oversaw these kinds of things called me in and asked me why I went to the WCB. Obviously I was naïve about such matters and responded as such. I was sent away with a warning that all medical problems be reported to the Safety Department first so they could investigate, then send me to the Company doctor. Shades of Silverwood. (See the movie with Cher. Cover-up. Cover-up.)
Friday, July 19, 2019
I heard a story about a chlorine spill that drifted across the complex towards the Herbicides Plant. Although the guys had Scott Air Pacs there to protect themselves from their own products, they didn’t plan on sticking around with this green cloud hanging over their plant. Some guys jumped into a pickup and headed for the gate. Whether the gas cloud was too thick, or they just didn’t care, they didn’t stop to open the steel gate. Crash!! Knocked that sucker flat to the ground. Gate? What gate?While working at Chlor-Alkali I got a sore throat (go figure!) so I went to my local doctor in St.Albert. I didn’t talk to the Company nurse or doctor because I didn’t see anyone else from the Chlorine Plant complaining, so assumed this was a "local" thing. The doctor asked where I worked and with what chemicals. As soon as I mentioned the word "chlorine" he said "Well that’s why you have a sore throat!". (Doctors go to school for 10 years to make this quick determination.) He then made me fill in a form for the Workmen’s Compensation Board. A week later, the Safety Supervisor who oversaw these kinds of things called me in and asked me why I went to the WCB. Obviously I was naïve about such matters and responded as such. I was sent away with a warning that all medical problems be reported to the Safety Department first so they could investigate, then send me to the Company doctor. Shades of Silverwood. (See the movie with Cher. Cover-up. Cover-up.)
Speaking of the Medical Facility….
Friday, July 19, 2019
As the complex grew, the necessity for better medicals and closer control became more important. So Dow built a small medical center in the administration building and staffed it with first one nurse, then two. They then began routine medical tests, mandatory for all employees. They did hearing tests, vision, usual blood pressure, cardiogram, etc. They also took blood, for analysis at the clinic in town. A very dangerous and unfortunate incident developed from some blood samples taken by the nurse one day. She got the names mixed up on the blood samples. Now just this fact alone would not have caused a problem had the blood work not turned up any difference between the two involved. But what happened was one of the men had a serious liver ailment that the other (healthy) man suddenly inherited! So the guy who had been "okay" suddenly got a serious disease, and the sick guy (who knew before this test he had a problem), was suddenly cured! Boy, did the fur fly over this one. All into the same fan as the other stuff.
Cell Operations
I had some pretty terrible jobs to do as cell operator. I hesitate to say it was the worst job at Chlor-alkali, because I would hang out with the other operators once in a while, and observe their jobs. I was satisfied that we all ALL had crappy jobs.
Friday, July 19, 2019
I was the cell operator at Chlor-Alkali. It was my job to take care of the brine well pumps and the cells. After I produced the raw products, it was up to the other operators (acid and caustic) to treat it and convert it to useful high grade product.I had some pretty terrible jobs to do as cell operator. I hesitate to say it was the worst job at Chlor-alkali, because I would hang out with the other operators once in a while, and observe their jobs. I was satisfied that we all ALL had crappy jobs.
Safety Considerations
Friday, July 19, 2019
One of my rotten jobs was taking samples. Every two hours, I had to carry a large container, that held forty 100 ml sample bottles, to the top of a chosen skid and sample each one of the forty lines going into each cell. The minimum required safety gear (now fashionably known as "Personal Protective Equipment") was hard hat, monogoggles, acid-proof high top rubber boots with 1" rubber soles, thick high temperature rubber gloves, and a respirator. The monogoggles completely protected the eyes by forming a seal around the user’s face, much like gas-welding goggles, or ski goggles. The lens was unbreakable polycarbonate, which also resisted scratches. The rubber boots weighed a couple pounds because they were steel shank and toe, in addition to covering half of the shin. These things were good for any abuse. Walk in mud, caustic, concentrated acids, with impunity. We were supposed to check for continuity of the boots before starting the shift, or any time we felt our toes tingle when walking on top a skid. We never forgot that there was only porous concrete separating our soles from 550 volts DC and 80,000 amperes. We would be reminded when we felt a "buzz" in the boot. The respirator was a mouth type, that is, a person stuck it in his mouth and put his lips over the rubber piece to get a good seal. Quite similar to the mouthpiece of underwater diving gear. At the end was a carbon canister, good for small concentrations of chlorine. It was no substitute for a Scott Air Pac, but would allow a person to work, and save himself in case of a bad leak. (Save himself for 10 seconds. We could hold our breath THAT long!)
Das Boot
One of the most irritating occurrences was when a feeder broke at the header. The brine would piss out 20 feet, getting the operator all wet in the process. Remember the brine is conductive at this point, and this solution is connected to that high voltage just below the operator. I was hit in the face more than once with this stuff, once when I had my monogoggles on my forehead! It was early in my career as a cell operator and I got careless, removing my goggles so I could "see" better. They got fogged up due to the extreme 35 degree heat and humidity on top of the skids. I got some of the solution in my eye, left the damn feeder to piss way up in the air, and ran down to the eyewash fountain. I was okay, and again didn’t report the incident for fear of being called an idiot. I wanted to go blind instead. (The brine contained about 10% by volume of Hydrochloric Acid). I was an idiot, who needed to be called one?
, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua.
Friday, July 19, 2019
The process of sampling sounds fairly simple, and it was: pull the plastic tube out of the cell (watch the green gas come up the little hole), and let the brine/acid solution fill the little bottle. Well by the time I’d get half-way through the 40 cells, my hands would start to tingle because my hands were sweating inside my insulated rubber gloves. Sometimes I would continue (after all it was just a little buzz) because I was too lazy to go get a fresh pair. When I got the "buzz in da’ boot", I couldn’t just change my boots because we only were issued one pair. I could change my socks, but that was too trouble-some. I could endure more in my feet than I could in my hands so I usually ignored it. It wasn’t painful, just irritating because you always wondered what would happen if you stepped on an area that was maybe a little thinner or more conductive than others.One of the most irritating occurrences was when a feeder broke at the header. The brine would piss out 20 feet, getting the operator all wet in the process. Remember the brine is conductive at this point, and this solution is connected to that high voltage just below the operator. I was hit in the face more than once with this stuff, once when I had my monogoggles on my forehead! It was early in my career as a cell operator and I got careless, removing my goggles so I could "see" better. They got fogged up due to the extreme 35 degree heat and humidity on top of the skids. I got some of the solution in my eye, left the damn feeder to piss way up in the air, and ran down to the eyewash fountain. I was okay, and again didn’t report the incident for fear of being called an idiot. I wanted to go blind instead. (The brine contained about 10% by volume of Hydrochloric Acid). I was an idiot, who needed to be called one?
, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua.

Getting back to this errant feeder, I had to get a
similar
piece of plastic tubing , remove the broken piece
with it’s
bushing and lock nut, and install these pieces on
the new
tube. Then, with 30 or 40 pounds of pressure
resisting my efforts to slip this nut back onto the
header,
I would get all wet again. Have you ever been
sprayed with a hose by your little brother and tried
NOT to
get wet by putting your hand in front of the nozzle that was pointed at you? That’s the
effect. Then I was really wet and salty, like sushi. Pickled sushi. I would finish my
sampling round before changing in case it happened again. Man, I just hated it when
that happened.
Hot Roddin’
It was important that the pH be maintained at somewhere between 1.35 and 1.5. The sampling procedure was a backup for the installed pH analyzers that failed at least once per shift. The analysis I did was quite quick, though not automated as much as it could have been, even back then in the ‘70s. Each of the 40 was analyzed, the date logged, and the SuperTech would look at them to see if he had to make a process, or pH meter calibration change. The pH meters actually were part of a feedback control loop, so if the pH dropped, less acid would be mixed in with the brine, and if the pH rose, more acid would be added. If the meter itself was giving a false reading, as determined by my analysis, then the ‘super’ could "re-zero" the analyzer to put things back in sync.
Friday, July 19, 2019
Another crappy job, related to the above was "rodding" a feeder. If the flow wasn’t up to par with the other ones, due to lines plugging up with salt, I had to take a metal welding rod and shove it into the feeder spewing out the acidized brine. Once again, shock city. And almost invariably, the brittle (from the heat and chemical mistreatment) plastic tube would snap and I had to replace it anyway. Arrrgghhhh!It was important that the pH be maintained at somewhere between 1.35 and 1.5. The sampling procedure was a backup for the installed pH analyzers that failed at least once per shift. The analysis I did was quite quick, though not automated as much as it could have been, even back then in the ‘70s. Each of the 40 was analyzed, the date logged, and the SuperTech would look at them to see if he had to make a process, or pH meter calibration change. The pH meters actually were part of a feedback control loop, so if the pH dropped, less acid would be mixed in with the brine, and if the pH rose, more acid would be added. If the meter itself was giving a false reading, as determined by my analysis, then the ‘super’ could "re-zero" the analyzer to put things back in sync.
Rodding Caustic Outlets
Rodding could cause another problem. If the rod hit the asbestos diaphragm it could knock a piece off and expose the metal cage underneath, and the graphite anode under that. Then the caustic flow would turn black forcing the operator into a quick action before the anode became consumed by the caustic. This membrane was all that separated the caustic from the brine, a kind of osmosis curtain. Next rotten job was mixing up a solution of powdered asbestos with water, in a 5 gallon pail. We did this on top of the skid because when water was added to asbestos it was heavy like cement. The first time I saw this done and then had to do it myself, the operator training me said "You should put your respirator on, because asbestos isn’t the best stuff to inhale". He also compared it to Fiberglas insulation, saying it was as "irritating". People wonder why I laugh when I see a room cleared if a little chunk of asbestos (actually cellulose) insulation falls from the ceiling.
I have since read a report that showed that cigarette smokers are 8 times more likely to develop lung cancer if exposed to asbestos, because the asbestos acts as a "promoter". I smoked. I asbestosed. Damn.
Anyway, this thick soup of asbestos was then poured down the center of the cell through a 2 inch hole in the top of the cell. Boy the chlorine sure spewed out of there! By virtue of the natural brine flow through the damaged membrane the asbestos would just fill and plug the hole. Goodbye and have a nice day.
Friday, July 19, 2019
Another hated job, which only had to be done once per shift, was rodding the caustic outlets. The outlets were made of 2" plastic and were continually dumping out a hot 10% solution of sodium hydroxide into the large collecting trough all along the side of the skid. Since the skids were put together like some kind of Lego, for easy tear-down and re-build, the concrete collectors had to be temporary as well. As a result the trough was made up of 10 or 20 foot sections of concrete that were pushed together and held in place with the same mastic that made the cells leak proof. When the flow would diminish from any outlet, it was necessary to insert a large rod, similar to re-bar, into this outlet to restore flow. Once again, shock city, only much more prevalent. Here I would change gloves often, and make sure my boots weren’t conductive. We had a tester where we put our boot in water and held on to a metal lead; if the indicator light went on, we "theoretically" couldn’t proceed to the cell area.Rodding could cause another problem. If the rod hit the asbestos diaphragm it could knock a piece off and expose the metal cage underneath, and the graphite anode under that. Then the caustic flow would turn black forcing the operator into a quick action before the anode became consumed by the caustic. This membrane was all that separated the caustic from the brine, a kind of osmosis curtain. Next rotten job was mixing up a solution of powdered asbestos with water, in a 5 gallon pail. We did this on top of the skid because when water was added to asbestos it was heavy like cement. The first time I saw this done and then had to do it myself, the operator training me said "You should put your respirator on, because asbestos isn’t the best stuff to inhale". He also compared it to Fiberglas insulation, saying it was as "irritating". People wonder why I laugh when I see a room cleared if a little chunk of asbestos (actually cellulose) insulation falls from the ceiling.
I have since read a report that showed that cigarette smokers are 8 times more likely to develop lung cancer if exposed to asbestos, because the asbestos acts as a "promoter". I smoked. I asbestosed. Damn.
Anyway, this thick soup of asbestos was then poured down the center of the cell through a 2 inch hole in the top of the cell. Boy the chlorine sure spewed out of there! By virtue of the natural brine flow through the damaged membrane the asbestos would just fill and plug the hole. Goodbye and have a nice day.

I had never worn a respirator before coming to Chlor-Alkali
and it required some practice. The part that was put in the
mouth fit in there like a hot-dog with edges: once it was in,
you had to wrap your lips around the edge of the tube.
Rookies like me had to use the nose clamp to make sure
we never breathed through the nose when in a gassy area,
but I just couldn’t get used to it. Eventually, after forgetting
the plug so many times, and then finding myself in a green
atmosphere (chlorine is green, and heavier than air)
breathing through my nose, I learned. Pavlov’s dog all over again.
Friday, July 19, 2019
One problem that developed with the respirators was the mouthpiece would get salty/acidy from laying on the front of my chest which was usually wet with brine. Put that in your mouth and suck it for awhile. Not pleasant.I was a cigarette smoker when I worked there, but I might have had only 1 cigarette the entire shift. Even a small whiff of chlorine that would not need a respirator to work with would get into the body’s breathing parts and linger there, making a cigarette taste like the worst chemical taste you could ever have. It was nauseating. Almost made me quit. Should have when I had such a good reason to.
Dow provided free milk to all the plants like they provided coffee. In Chlor-Alkali it was a health thing, the other plants only got the milk because the chlorine guys did. Apparently the milk was good for those that were continually breathing in chlorine. I’m not sure if we had to inhale the milk or what. In any case, I drank the stuff.
Turn, Turn, Turn
At about the time I left Chlorine for EO, a new Chlor-Alkali plant was being constructed. This was a 600 ton-per-day chlorine producer, whereas the old plant where I worked was max’ed out at 300 tons per day. I do believe there is an even bigger plant than this, Chlor-Alkali III, that was built 10 years ago. Hey, I’m not writing a technical book here, I don’t feel I have to do research to get the facts!
At the chlorine plant, after my Cell Operator training , my immediate supervisor was the Supervising Technician, or as they were known at the time, the "Control Operator". This guy, who’s name I’ll never remember, and that’s unfortunate, was a really good guy to work with and for. He was always giving me helpful hints, asking me how my part of the plant was going, how my job was, etc. He would even come out and help me every once in a while. What I remember most about him was that he was always cheerful and never really pushed his weight around in the control room. He left Dow a few years later to work up in a new pulp and paper mill in Grande Prairie, because there was possibility of management type jobs up there if you got in early enough.
(Addendum April 2006:Actually I do remember. His name was Mike Samoil)
Friday, July 19, 2019
I only had to work one turn-around on day-shift. A turn-around was a big shut-down which we had about once every year. All through the year anything that needed repair that couldn’t be repaired while the plant was running was scheduled for the turn-around. Since the cell area was mine to take care of, I was the one responsible for making equipment safe for the workers to work on. This involved closing valves, draining pipes, purging with water or nitrogen, locking out electrical equipment so it wouldn’t start up. After turn-around was the startup, which was equally as harrowing because of all the work involved. Brine levels had to be brought up properly, in sync with power increases. As the caustic started to flow, there was lots of rodding to do. Sometimes 20 of the 40 cells, on ONE skid would have to be rodded. And during startup there were always chlorine releases into the building, which is why only the cell operator was allowed in the area at this critical time. (When I left Dow, I forgot to thank them for their consideration…)At about the time I left Chlorine for EO, a new Chlor-Alkali plant was being constructed. This was a 600 ton-per-day chlorine producer, whereas the old plant where I worked was max’ed out at 300 tons per day. I do believe there is an even bigger plant than this, Chlor-Alkali III, that was built 10 years ago. Hey, I’m not writing a technical book here, I don’t feel I have to do research to get the facts!
At the chlorine plant, after my Cell Operator training , my immediate supervisor was the Supervising Technician, or as they were known at the time, the "Control Operator". This guy, who’s name I’ll never remember, and that’s unfortunate, was a really good guy to work with and for. He was always giving me helpful hints, asking me how my part of the plant was going, how my job was, etc. He would even come out and help me every once in a while. What I remember most about him was that he was always cheerful and never really pushed his weight around in the control room. He left Dow a few years later to work up in a new pulp and paper mill in Grande Prairie, because there was possibility of management type jobs up there if you got in early enough.
(Addendum April 2006:Actually I do remember. His name was Mike Samoil)
Monty
Friday, July 19, 2019
The guy who trained me for his position of cell operator, Monty, moved one supposed "step up", to become the Acid Operator. I say "supposed" because Monty had to take care of the process whereby hydrogen gas was lit in a burner and allowed to burn in an atmosphere of chlorine gas. This was done in a tower with little "waterfalls" designed to absorb the combination of the gases, creating hydrochloric acid (HCL). He was kind of like a bull in a china shop: everything he did, he did it with total abandon. (As the driver of the "trackmobile", a small railroad engine for moving tank cars around, he derailed it and a string of cars more than once. He actually ran over the derailer.) When he taught me my job, it was like learning how to sweep floors: "do this, do that, simple, don’t worry, blah, blah, blah". He was talking about dealing with chlorine gas, hydrogen gas, caustic soda, hydrochloric acid, like he was talking about roasting marshmallows! He knew his job and he wasn’t afraid of anything. I was afraid at first, but got used to the idea of what I was dealing with. I attribute my good safety record to just that: being afraid. I always respected things that I knew would kill me, and not feel remorse in the morning!
Getting Initiated CA1 shower
Friday, July 19, 2019
Since I was the only operator at Water Treat on my shift, I never had any opportunities to pull tricks on others. In fact, because I was so new to industrial work, I never knew that horse-play even took place. That changed when I moved to the Chlor-Alkali (also known as Chlorine) Plant where there were 4 operators on shift. On my very first day in my new job there, I was taking a hot shower after a miserably long 12 hour night shift, when another operator comes bursting into the shower stall with a 5 gallon pail of cold water. This had the effect of waking me up, and was my initiation into the group. I heard from others that I was lucky I was wet when I got hit. Others had been doused just as they got their work clothes on at the beginning of the shift, and still others, with a bucket over the toilet stall!
Gas Bags
Friday, July 19, 2019
Another pastime at the Chlorine Plant that others took part in was filling paper bags with hydrogen gas, a product we produced there in abundance. The bags were filled and allowed to float up in the air. Well, one time one of these bags ventured close to a stack that was burning excess hydrogen and the bag started on fire. This was definitely a dangerous situation, but luckily the bag burned itself out before something else caught fire. The practice ended that night.
The Big Sausage
Friday, July 19, 2019
Another time, one of the senior operators was having fun with "surgical hose", the kind of soft and pliable rubber hose used to hook chemical apparatus together. He had wired one end of a 2 foot section, closed and attached the other end to the lab water tap, the type with a pointed nozzle. As he slowly turned the tap on, the hose began to expand, both radially and along it’s length: it was almost 3 inches in diameter and over 5 feet long and still expanding! By then no one wanted to be around it in case it "blew" when one got close. So we all moved to the other side of the control room to watch. Just then, the weight of the "sausage" pulled the end out of the tap and all the water shot out of the open end, mostly into the sink. What a let-down! Just before we left our shift, the operator hooked it up again, but tied it off at the tap so it wouldn’t slip off. I never did find out if the next shift found it in time, or if they were "surprised". Heh heh heh.
Field of Dreams
Friday, July 19, 2019
The process of making chlorine involved the hydrolysis of brine which, using immense amounts of power. This created an immense magnetic field, so intense that we were told never to wear a watch, because it wouldn’t work the next day. One experiment that I performed to measure the power of this field was hanging tools from my tool belt on the side of the chlorine cell. I could take my 12" crescent wrench and have it hang horizontally from the vertical surface of the cell! That’s right, at right angles, and it would stay there! I also tried nails and made a long chain with them, the same way.
Mama Was Poppin’
Friday, July 19, 2019
My wife was in labour with my son one midnight shift that I was working. Unlike the kind of jobs most people work where they can just leave and no one will miss them, I could not just leave. My job was very important and we just couldn’t phone someone at 2 AM to ask him to come in and work. When my shift ended, I drove right to the hospital, spent all day with my wife who was still in labour. Finally, after 13 hours the boy was born, and after another hour with the wife, I left. I slept for about 2 hours at my mother’s, got some hand-outs for my lunch that night, then went back to work for another 8-hour midnight shift. Nowadays, wimpy fathers take "paternity" leave for two weeks.